Yesterday I wrote briefly about the ongoing release of methane from the crust of Mars. This is the topic of today’s Astronomy Picture of the Day:
Here’s the description from APOD:
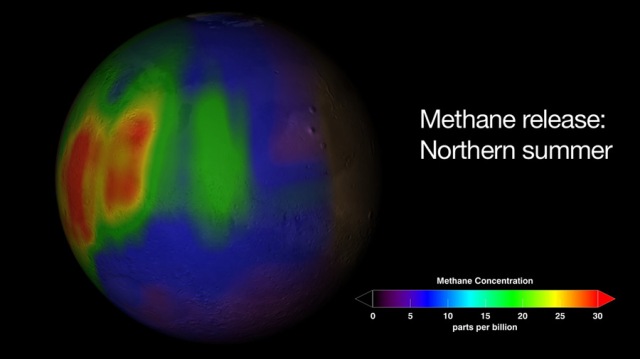
Why is there methane on Mars? No one is sure. An important confirmation that methane exists in the atmosphere of Mars occurred last week, bolstering previous controversial claims made as early as 2003. The confirmation was made spectroscopically using large ground-based telescopes by finding precise colors absorbed on Mars that match those absorbed by methane on Earth. Given that methane is destroyed in the open martian air in a matter of years, the present existence of the fragile gas indicates that it is currently being released, somehow, from the surface of Mars. One prospect is that microbes living underground are creating it, or created in the past. If true, this opens the exciting possibility that life might be present under the surface of Mars even today. Given the present data, however, it is also possible that a purely geologic process, potentially involving volcanism or rust and not involving any life forms, is the methane creator. Pictured above is an image of Mars superposed with a map of the recent methane detection.
This discovery was actually made using Earth-based instruments, not instruments on orbiters. Scientists are already looking into means of determining the isotopic composition of the methane (the ratio of carbon-12 to carbon-13) to gain additional clues as to whether this methane is produced by biological or geological means. Biological processes fractionate carbon isotopes in a slightly different ratio than non-biological processes, at least on Earth. Isotope fractionation occurs because molecules having slightly different mass have slightly different physical properties. For example, heavy water (H2O made with hydrogen-2) has a slightly different boiling point than ordinary water (made with hydrogen-1). Likewise, methane made with carbon-13 has slightly different properties than methane made with the more abundant carbon-12.
Mass spectrometers, the instruments used to measure isotope ratios, are too heavy at present to place on landers. There are, however, spectrographic methods that can be used to determine isotopic ratios, and it is possible that these instruments could be placed on future probes to Mars.
Grace and Peace
